|
Protocol
for RNA Quantitation Using RiboGreen�
1.
INTRODUCTION
RiboGreen®
RNA Quantitation Reagent is an ultra-sensitive fluorescent
nucleic acid stain. It is a simple and rapid procedure
for measuring RNA concentration in solution for several
molecular biology procedures. A few such procedures
include in vitro transcription, Northern
blot analysis, reverse transcription PCR, differential
display PCR, S1 nuclease assays, RNAse protection assays
and cDNA synthesis for library production.
The
determination of absorbance at 260nm is the most commonly
used technique for measuring RNA concentration. Major
disadvantages of this method are poor sensitivity (4
µg/ml RNA in solution) and interference in signal
levels from contaminating components such as nucleotides,
proteins and salts in the RNA solution. The use of an
ultra-sensitive fluorescent nucleic acid stain such
as RiboGreen® alleviates these problems.
The
Turner Designs dual-channel Picofluor™ fluorometer
along with the Molecular Probes RiboGreen®
RNA Quantitation Kit allows rapid and accurate measurement
of RNA concentration in solution. The RiboGreen®
reagent is non-fluorescent when free in solution. Upon
binding RNA, the fluorescence of the RiboGreen®
reagent increases more than 1000-fold. The RNA
bound RiboGreen® reagent has an excitation
maximum of ~500nm and an emission maximum of ~525nm
similar to Fluorescein.
The
linearity of the assay is maintained even in the presence
of contaminants in the RNA solution. RiboGreen®
also binds DNA. RNA-DNA mixed samples may be pretreated
with DNAse to generate an RNA selective assay. As little
as 2.5ng/mL RNA can be quantitated using the blue optical
configuration of the Picofluor™. The RiboGreen®
assay is ~200-fold more sensitive than ethidium
bromide-based assays and ~1000-fold more sensitive than
absorbance measurements at 260 nm. The linear dynamic
range extends over 4 orders of magnitude from 2.5 ng/mL
to 1 µg/mL RNA (Figure 1) using two different
concentrations of the RiboGreen® reagent.
A linear dynamic range of 10ng/mL to 1µg/mL can
be achieved by the use of a high concentration of the
dye. Furthermore, by the use of a lower concentration
of the dye, a lower range of 2.5ng/mL to 50ng/mL can
be measured.
2.
MATERIALS REQUIRED
- Picofluor™
Fluorometer with blue optical configuration (P/N 8000-003
or 8000-004).
- 10
mm X 10mm Polystyrene cuvettes (P/N 7000-957).
We recommend these cuvettes in particular. We found
that variations between replicates decreased markedly,
leading to better detection limits with these polystyrene
cuvettes.
RiboGreen® RNA Quantitation Kit, (R-11490)
Molecular Probes, Inc.
The kit contains :
- 1
mL of RiboGreen® RNA quantitation reagent
-
25 mL of 20X TE assay
- 5
200 µL aliquots of 100 µg/mL 16S and 23S
ribosomal RNA standard (from E. coli), in TE
buffer.
Handling,
storage and the use of the reagents should be performed
in accordance with the product information sheet supplied
by Molecular Probes, Inc.
- Nuclease-free
water (see Section 3.1, below)
3.
EXPERIMENT PROTOCOL
3.1
Assay Buffer Preparation
TE
assay buffer (10 mM Tris-HCl, 1 mM EDTA, pH 7.5) is
used for diluting the RiboGreen® reagent
and the RNA samples. It is imperative that the TE
buffer is free of contaminating nucleases and nucleic
acids. Clean disposable gloves should be worn during
handling and preparation of all materials and solutions.
All solutions should be prepared in sterile disposable
plastic ware or nuclease-free glassware, using nuclease-free
pipettes.
The
20X TE buffer that is included in the RiboGreen®
RNA Quantitation Kit is nuclease-free and nucleic
acid-free. This buffer is also available from Molecular
Probes, Inc. as a separate item (catalog number T-11493).
Prepare the 1X TE working solution by diluting the
concentrated buffer 20-fold with nuclease-free water.
Nuclease-free
water should be prepared by treating distilled, deionized
water with 0.1% diethylpyrocarbonate (DEPC), incubating
for several hours at 37°C and autoclaving for
at least 15 minutes at 15-lbs/sq. inch to sterilize
and eliminate DEPC. Caution: DEPC is a suspected carcinogen
and should be handled with care. Compounds containing
amines, such as Tris, will react rapidly with DEPC
and should be added to DEPC treated water only after
DEPC is removed by heating. Removal of DEPC by heating
is also important to prevent carboxyethylation of
the RNA sample. Alternatively, commercially available
Nuclease-free water may be used.
3.2 Reagent Preparation
The
RiboGreen® RNA Quantitation Reagent
is supplied as a 1-mL concentrated dye solution in
anhydrous dimethylsulfoxide (DMSO). The DMSO stock
solution should be stored at -20°C. Allow the
reagent to warm to room temperature.
To
measure levels between 1000 -10 ng/mL RNA, prepare
the high range assay reagent by making a 1:200 dilution
of the concentrated RiboGreen® Reagent
into TE. A dilution of 100µL RiboGreen®
Quantitation Reagent to 20.0 mL TE is an adequate
working solution to assay 20 samples in 2mL volumes.
To
measure levels between 50 -2.5 ng/mL RNA, prepare
the low range assay reagent by making a 1:2000 dilution
of the concentrated RiboGreen® Reagent
into TE. A dilution of 10µL RiboGreen®
Quantitation Reagent to 20.0 mL TE is an adequate
working solution to assay 20 samples in 2mL volumes.
Prepare
these solutions in a plastic container, as the reagent
may adsorb to glass surfaces. The working solution
of the RiboGreen® Reagent must be protected
from light with foil or placing it in the dark to
prevent photo degradation. For best results, this
solution should be used within a few hours of its
preparation.
3.3
RNA Standard Curve
1.
Prepare a 2µg/mL solution of RNA in TE using
nuclease-free plastic ware. Determine the RNA concentration
on the basis of absorbance at 260 nm (A260) in a cuvette
with a 1 cm pathlength; an A260 of 0.05 corresponds
to 2 µg/mL RNA.
2.
The 16S and 23S ribosomal RNA standard, provided at
100 µg/mL in the RiboGreen® RNA
Quantitation Kit, can simply be diluted 50-fold in
TE to make the 2 µg/mL working solution. For
example, 40 µL of the RNA standard mixed with
1.96 mL of TE will be sufficient for the standard
curve described below in Table 1.
3.
For the high-range standard curve, dilute the 2 µg/mL
RNA solution into disposable cuvettes as shown in
Table 1. For the low-range standard curve, first dilute
the 2 µg/mL RNA solution 20-fold with TE buffer
to make a 100 ng/mL RNA stock solution and use this
to prepare the dilutions shown in Table 2.
4.
Add 1.0 mL of the appropriate aqueous working solution
of RiboGreen® reagent (prepared in
Section 3.2) to each cuvette. The high-range working
reagent should only be used for performing the high-range
assay and the low-range working reagent should only
be used for performing the low-range assay. Mix well
and incubate for 2 to 5 minutes at room temperature,
protected from light.
5.
Set-up the PicoFluor™ fluorometer as per
instructions in the User's manual. Power up the instrument
by pressing the [ON/OFF] button. Use the [A/B] button
to toggle to the "Blue" channel. Press [STD
VAL] to program in the concentration of your calibration
standard. Use the up and down arrows to change the
concentration value. Hold down the arrow key to activate
faster scrolling. When ready, press the [CAL] button
to start the calibration. The Picofluor's screens
will lead you through the calibration process.
6.
Measure the fluorescence of the remaining standards
to generate a standard curve of fluorescence versus
RNA concentration.
|
Vol.(uL)
2ug/mL RNA stock
|
Vol.
(uL) of TE
|
Vol.
(uL) diluted RiboGreen® reagent
|
Final
RNA concentration in RiboGreen®
assay
|
| 1000 |
0 |
1000 |
1000
(ng/mL) |
| 750 |
250 |
1000 |
750
(ng/mL) |
| 500 |
500 |
1000 |
500
(ng/mL) |
| 250 |
750 |
1000 |
250
(ng/mL) |
| 100 |
900 |
1000 |
100
(ng/mL) |
| 50 |
950 |
1000 |
50
(ng/mL) |
| 25 |
975 |
1000 |
25
(ng/mL) |
| 10 |
990 |
1000 |
10
(ng/mL) |
| 0 |
1000 |
1000 |
0
(ng/mLl) |
Table
1. Protocol for preparing high range standard curve.
|
Vol.(uL)
100 ng/mL RNA stock
|
Vol.
(uL) of TE
|
Vol.
(uL) diluted RiboGreen® reagent
|
Final
RNA concentration in RiboGreen®
assay
|
| 1000 |
0 |
1000 |
50
(ng/mL) |
| 600 |
400 |
1000 |
30
(ng/mL) |
| 500 |
500 |
1000 |
25
(ng/mL) |
| 400 |
600 |
1000 |
20
(ng/mL) |
| 300 |
700 |
1000 |
15
(ng/mL) |
| 200 |
800 |
1000 |
10
(ng/mL) |
| 100 |
900 |
1000 |
5 (ng/mL) |
| 50 |
950 |
1000 |
2.5
(ng/mL) |
| 20 |
980 |
1000 |
1
(ng/mL) |
| 0 |
1000 |
1000 |
0
(ng/mL) |
Table
2. Protocol for preparing low-range standard curve.
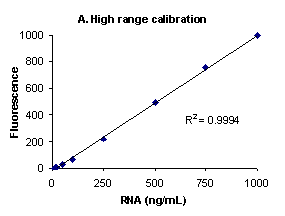
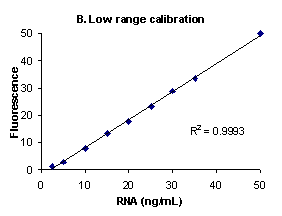
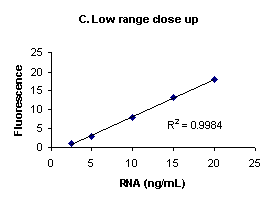
Figure 1. High range (A), low range (B) and low
range close up (C) E. coli ribosomal RNA standard
assays performed using RiboGreen® RNA
quantitation reagent and the PicoFluor™
handheld fluorometer. Sensitivity calibrations were
performed separately for the high and low assay ranges.
3.4 Sample Analysis
1.
Dilute each experimental RNA solution in TE to a final
volume of 1.0 mL in disposable cuvettes or test tubes.
It may be useful to prepare several dilutions of each
experimental sample. For example, if a series of RNA
samples contain widely differing salt concentrations,
they cannot be compared to a single standard curve.
To avoid this problem, simply adjust the concentration
of contaminants to be the same in all samples, if
possible. See Section 3.5 for information on eliminating
DNA from the sample.
2.
Add 1.0 mL of the appropriate RiboGreen®
reagent (prepared in Section 3.2) to each sample.
Incubate for 2 to 5 minutes at room temperature, protected
from light.
3.
Measure the fluorescence of each sample using the
same instrument calibration conditions as used to
generate the standard curve (see Section 3.3).
4.
If the standard curve has been constructed from background-subtracted
data (see Section 3.3), subtract the reagent blank
fluorescence reading from that of each of the samples.
5.
Determine the RNA concentration of each sample from
the standard curve you generated in Section 3.3.
3.5 Eliminating DNA from Samples
RiboGreen®
reagent also binds to DNA. The following procedure
for pre-treating the sample with RNase-free DNase
can eliminate this background fluorescence:
1.
Prepare 10X DNase digestion buffer: nuclease-free
200 mM Tris-HCl, pH 7.5, containing 100 mM MgCl2
and 20 mM CaCl2.
2.
Add 0.11 sample volume of 10X DNase digestion buffer
to each DNA-containing sample (for example, to a 9
mL sample, add 1 mL 10X buffer).
3.
Add about 5 units of RNase-free DNase I per µg
of DNA thought to be in the sample.
4.
Incubate the sample at 37°C for 90 minutes.
5.
Dilute the sample 10X in TE to keep the effects of
the digestion buffer salts to a minimum.
6.
Perform the RiboGreen® assay.
4.
REFERENCES
1.
Anal. Biochem. 17, 100 (1966).
2. Anal. Biochem. 265, 368 (1998).
3. Molecular Cloning: A Laboratory Manual, Second
Edition, J. Sambrook, E.F. Fritsch and T. Maniatis,
Cold Spring Harbor Laboratory Press, Cold Spring Harbor,
New York (1989).
5.
WARNINGS AND PRECAUTIONS
The
RiboGreen® RNA Quantitation Reagent
is the subject of patent applications filed by Molecular
Probes, Inc. and is not available for resale or other
commercial uses without a specific agreement from
Molecular Probes, Inc. RiboGreen® is
a registered trademark of Molecular Probes, Inc.
| 
